Synapses modify the neuronal membrane potential. The strength and time course of synaptic influence, together with the internal membrane properties of the neuron, ultimately determine the "spike times" of action potentials. Some neurons don't use action potentials, instead they transmit graded responses along their membranes, but these neurons can still be influenced synaptically. The word "synapse" in a general sense is meant to include all forms of local connectivity, for example gap junctions, but historically it refers specifically to a vesicular form of transmission.Vesicular TransmissionThe classic synapse is unidirectional, from an axon terminal to the dendrite of another neuron. The presynaptic axon terminal is filled with vesicles containing neurotransmitter, which diffuse across the synaptic cleft to interact with receptors on the postsynaptic side. Synapses can be made anywhere, on the dendrites, on the cell body, even on other axons. There are many specialized kinds of synapses in a human brain, for instance the glomeruli in the cerebellum. The first figure below stylizes an oversimplified "typical" synapse, showing the release of neurotransmitter from synaptic vesicles. The second figure shows the fusion of transmitter-loaded vesicles with the axon terminal, and the expulsion of neurotransmitter into the synaptic cleft. Fusion of vesicles with the presynaptic membrane depends on calcium, which enters the axon terminal during an action potential. When calcium enters the synapse, it causes the vesicles to fuse with the presynaptic membrane, releasing their contents into the synaptic cleft, which is a tiny gap between the pre- and post-synaptic membranes maintained by specific protein scaffolding. After release from the vesicles, the neurotransmitter diffuses across the cleft and binds with receptors on the postsynaptic side. After it's done binding, it can be either degraded or recycled (and either process may involve active reuptake, into neurons and/or glial cells - the reuptake is accomplished by specialized proteins called "transporters").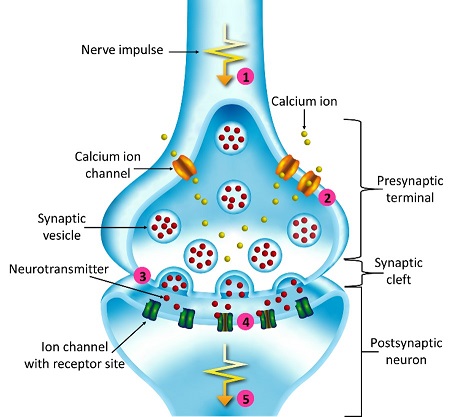 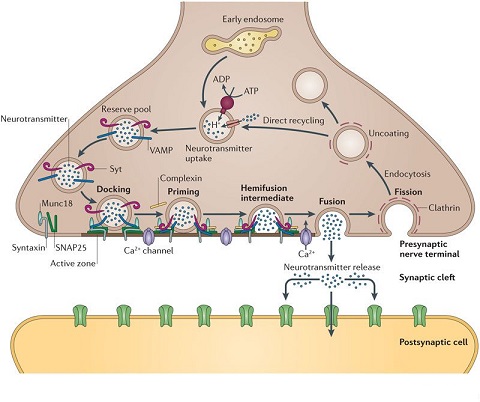 Neurotransmitter filled vesicles are synthesized and packaged in the cell body, and transmitted down the axon to the synaptic terminals by a network of microtubules related to the cytoskeleton. This process is regulated, it can be sped up or slowed down according to demand. There is both anterograde transport and retrograde transport, used vesicles are transported back to the cell body where they can be refilled. In conditions of changing demand, the quantal transmission hypothesis specifies that the amount of neurotransmitter per vesicle remains relatively constant, but the number of available vesicles can change. In the figure below MTOC is a microtubule organizing center.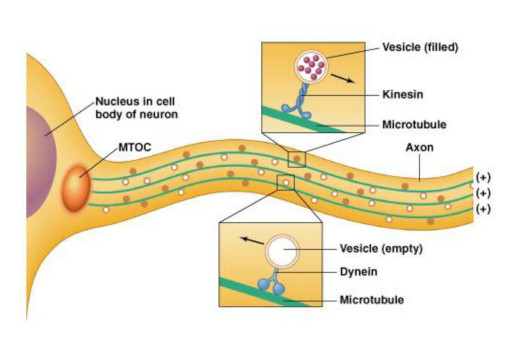 The totality of the synapses emanating from a neuron may be quite diverse. Some neurons use more than one transmitter. The axons branch and send collaterals all over the brain, and in some areas the receptors may be different and have unique postsynaptic effects. However in most neurons, the branching patterns are very specific, and in some cases very local. In the cerebral cortex for example, the axons of lower layer cells may turn upward and branch to synapse within a narrow cylinder about equivalent in width to a mini-column. 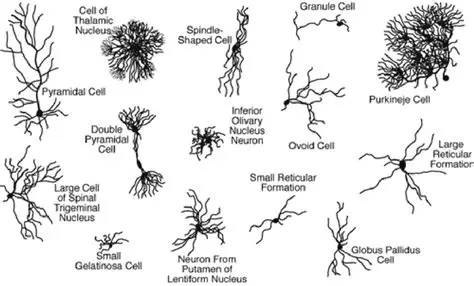 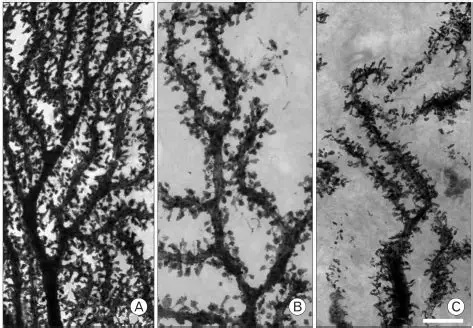 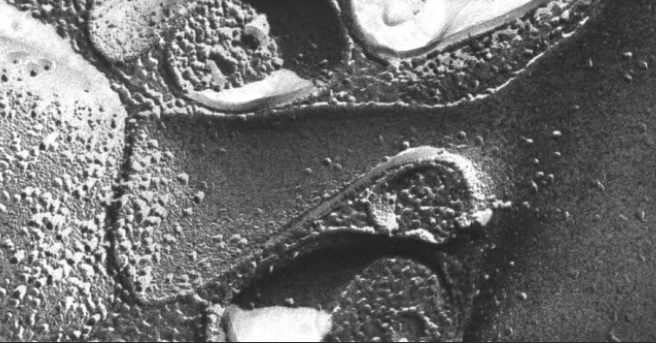 Spiny synapses are ubiquitous in the human brain. Here they are in the hippocampus and cerebral cortex, and they occur as well in the basal ganglia and cerebellum and many other subcortical structures.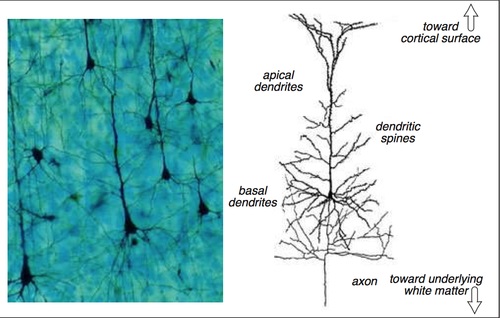 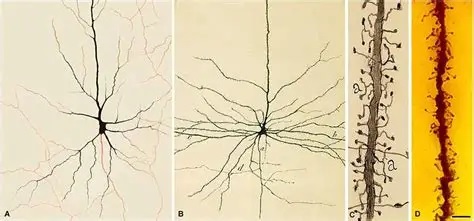 The synaptic spines are important for many reasons. One of the most important is isolation, which is necessary for synapses to be updated independently. Another reason is that each patch of spiny membrane can sustain independent subthreshold membrane oscillations. In effect, each spine can become its own oscillator, coupled to others by conductances along the membrane, but only weakly and controllably coupled because of the high impedance of the spine stalk and its biochemical compartmentation relative to the rest of the dendrite. We will revisit this scenario in more detail later, when we talk about the behavior of coupled oscillators. Below is the bursting behavior of a pyramidal neuron in the cerebral cortex, elicited by applying glutamate to the basal dendritic tree. You can see the small subthreshold membrane oscillations that occur even in the down state. These are often related to membrane resonances which in turn depend on the conductances and are therefore synaptically modifiable. Changes in the membrane time constant are one way to control the interval between spikes.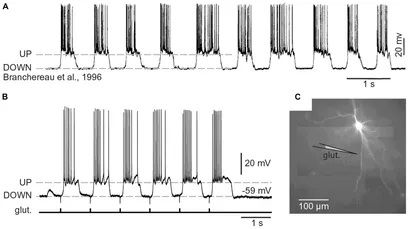 Synaptic activity results in excitatory and inhibitory postsynaptic currents. The time course of such currents could be anywhere from a millisecond to a second or more, depending on the kinetics of the underlying neurotransmission. There is typically a small delay associated with synaptic transmission, perhaps on the order of a millisecond or so.  The effect of incoming EPSP's may change when the postsynaptic membrane is driven into a more excitable state. This is especially true for multistable neurons that have a judicious combination of conductances that allow them to maintain multiple resting potentials depending on the time course of activity. For instance there are "plateau potentials" that are larger than ordinary EPSP's, and these are thought to be created by calcium conductances in the dendrites. The plateau potentials can put the neuron cell body into a different resting state, where it responds differently to incoming synaptic signals. Below are some of the plateau potentials related to dendritic spiking activity in neostriatal medium spiny cells. The plateau potentials typically result in bursts of spiking. They are compared with ordinary EPSPs that may result in one or a few spikes. Plateau potentials are different from dendritic spikes. They represent a stable equilibrium state of the neuronal membrane, whereas spikes are unstable and transient. Note the lengthy time course of the plateau in the figure. Plateaus are thought to put the neuron into a high-transmission mode, where reliability and throughput are increased and bursting is likely.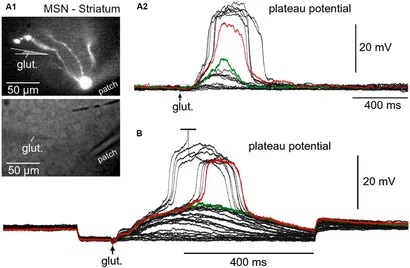 Gap JunctionsIn addition to the well known chemical synapses there are electrical synapses all over the brain, among neurons and among glia, and between neurons and glia. A gap junction is a low resistance electrical pathway between neighboring cells. Gap junctions are regulated dynamically, and the signaling delay through a gap junction can vary from nearly the membrane time constant, to much longer than a synaptic delay. Electrical transmission through a gap junction can be considerably faster than vesicular synaptic transmission, even on the order of microseconds rather than milliseconds. However typically there are diffusion effects related to geometry and transmission effects related to gap junction regulation, and the delay through a gap junction can reach 40 msec or more - which is slow compared to a 1 msec synapse but still within the generalized time frame of synaptic action. Gap junctions allow small molecules to pass, including ions and nutrients, and are important for homeostasis. (Goodenough & Paul 2009, Gansert et al 2008, Riol et al 2021)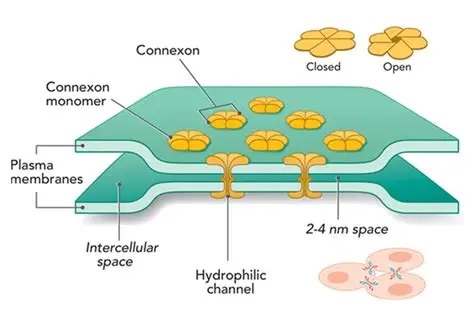 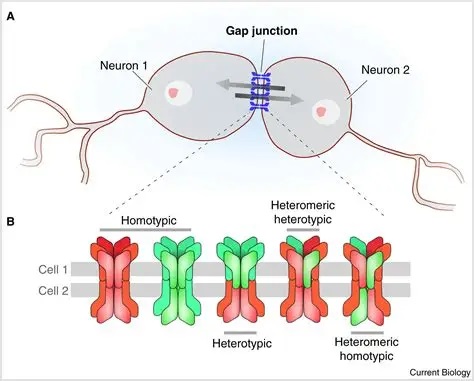 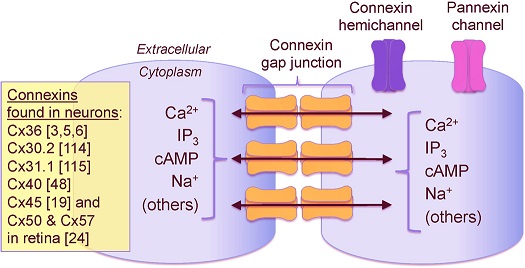 An interesting property of some gap junctions, useful for computation, is that they are rectifying. Depolarizing signals (but not hyperpolarizing signals) preferentially travel in one direction only, and hyperpolarizing signals (but not depolarizing signals) preferentially travel in the other direction only. The classic example is the escape circuit in the crawfish (Furshpan and Potter 1959). Second MessengersIn many cases the postsynaptic receptor has downstream effects in addition to its immediate interactions with ion channels. In some cases there are no ion channels at all, and synaptic effects are carried downstream by "second messenger" molecules like cyclic AMP and cyclic GMP. In some cases there are both ion channels and second messengers. The general mechanism of a second messenger is shown in the figure. As a rule, second messengers are quite slow compared to ligand gated ion channels.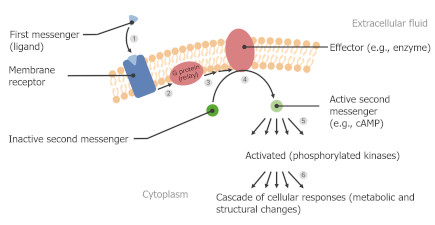 Generally, receptors can be classified into 4 broad types. There are ligand gated ion channels, G-protein coupled receptors, enzyme coupled receptors, and intracellular receptors. The ligand gated channels are what we've already discussed - when the neurotransmitter binds to the receptor the result is an opening or closing of one or more ion channels. The G-protein coupled receptors work differently, instead of altering an ion channel the result is an increase or decrease in the intracellular level of a "second messenger", frequently cyclic AMP (cAMP) or cyclic GMP (cGMP). The second messenger then has downstream effects, which may include a host of cytosolic and even nuclear interactions. In the third type of receptor (enzyme coupled), there is a direct action on intracellular proteins that control other processes (in other words, there is no second messenger between them). Such an action may include the binding or unbinding of a receptor from the cytoskeleton, thereby either locking it into place or leaving it free to diffuse in the membrane.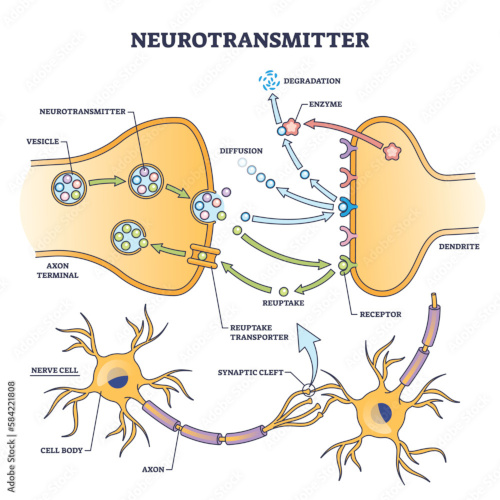 In the case of glutamate, the most common excitatory neurotransmitter in the brain, a single synapse may contain both ionotropic and metabotropic receptors, with different time courses and different actions. For example it is likely that the metabotropic receptors using second messengers regulate the ionotropic receptors, over time courses longer than the duration of an action potential. Most of the glutamate synapses in the brain are plastic, they exhibit a range of positive and negative changes in strength over time. We'll talk about plasticity separately (it's a lengthy topic).Synaptic WebIn the synaptic cleft between the pre- and post-synaptic membranes is often found a "synaptic web" that includes adhesion molecules that hold receptors in place and serve as docking sites for synaptic vesicles. In general the molecules around a synapse include a presynaptic density (possibly related to docking sites for transmitter-filled vesicles), a postsynaptic density (possibly related to localized concentrations of neurotransmitter receptors), and a network of interconnected structures in the synaptic cleft itself. An example is shown in the figure.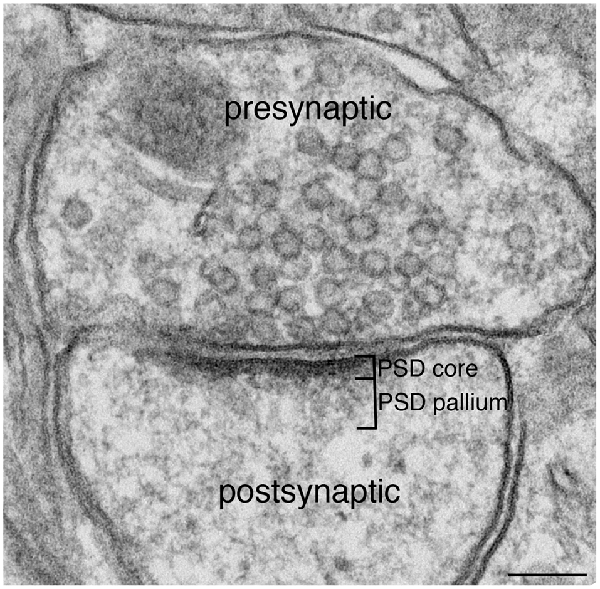 (figure from Dosemeci et al 2016) 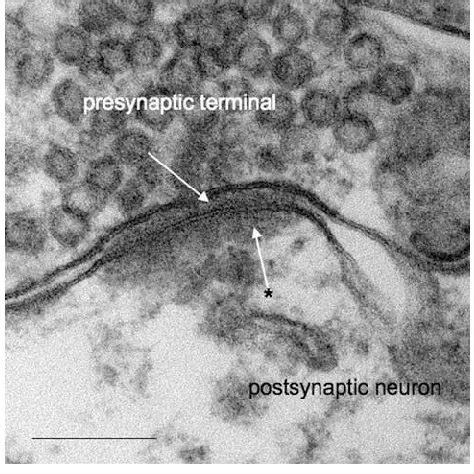 (figure from Dresbach et al 2008) 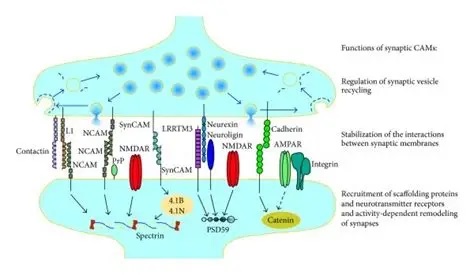 (figure from Leshchyns'ka and Sytnyk 2016) In addition to the synaptic web there is usually a noticeable continuation of the post-synaptic density into microtubules that end near the endoplasmic reticulum, especially in spiny synapses.Control of Transmitter LevelsNeurotransmitters are actively synthesized in the endoplasmic reticulum in the cell body of neurons, then packaged into vesicles in the Golgi apparatus, and transported down the axon to the synaptic terminals by an array of microtubules, where they are released by calcium-driven exocytosis. (Bentley and Banker 2016, Nakagawa 2024). This process is shown schematically in the figures.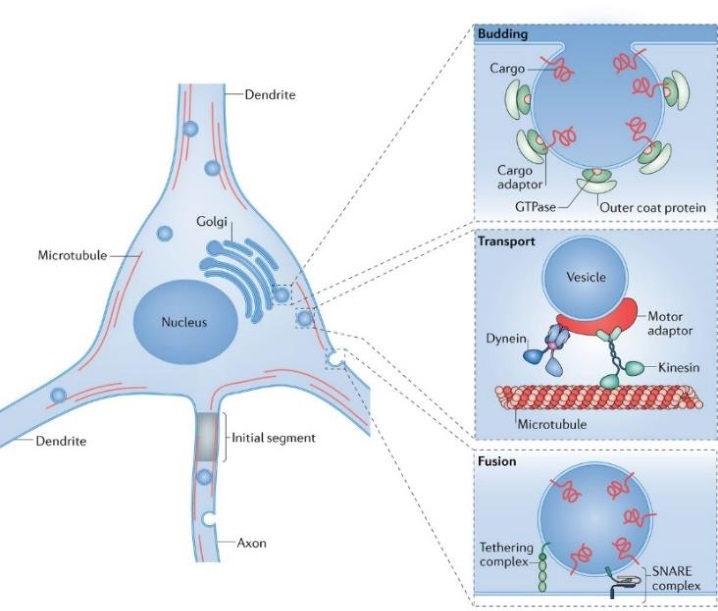 (figure from Bentley and Banker 2016) 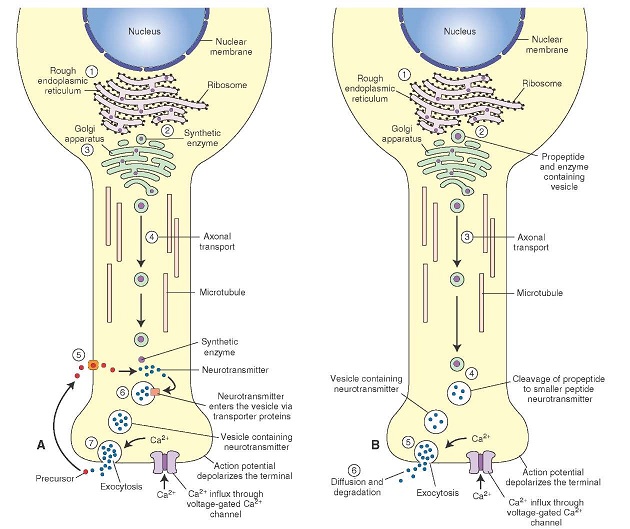 How does the cell regulate the amount of neurotransmitter that is available to be released? As shown above, transmitter-filled vesicles can come from two places, they can be freshly synthesized or they can be the product of recycling. The sum of these two sources results in a pool of available transmitter-filled vesicles in the synapse (you can see the pool of vesicles ready to be released in several of the electron micrographs above). Generally, the amount of transmitter packaged into a vesicle is constant, whereas the number of vesicles can vary. There is a condition of "synaptic exhaustion" where the production of vesicles can not keep up with the demand, and it is important to note that other molecules besides neurotransmitter (including mRNA) are transported in vesicles, so a condition of depletion will affect those concentrations as well. In some cases, especially in proximity to dendritic spines, there may be endoplasmic reticulum in the dendrites themselves, and it may contribute to local transmitter synthesis and especially to the local production of micro-RNA and specific proteins (Carvalhais et al 2026). Control of Receptor Levels and PositionsNeurotransmitter receptor levels are also carefully controlled throughout the brain. The strength, or "weight", of a synapse is determined by the ratio of neurotransmitter to postsynaptic effect. Many neurons have regulatory mechanisms that set the number of receptors, and the setpoint can vary with several types of synaptic plasticity. In particular, much is known about the various glutamate receptors in the brain (glutamate being the most prevalent excitatory neurotransmitter), and the ways they contribute to synaptic plasticity. The figure shows some varieties of glutamate receptors.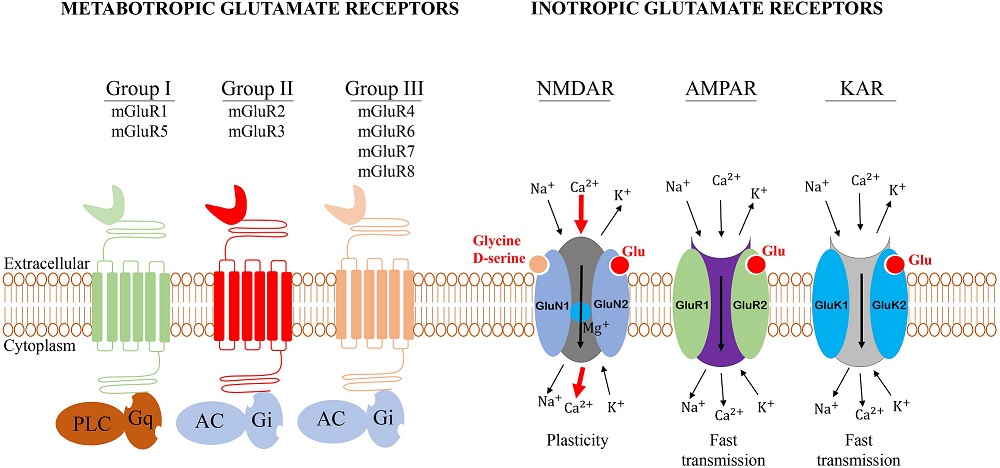 Generally in neurons there is control over receptor production (synthesis levels), receptor transport (the migration into synapses and spines), receptor location (in cooperation with the cytoskeleton), receptor binding (including postsynaptic effects like second messengers), and receptor release and degredation (possibly involving recycling, with or without vesicles, and possibly also involving glial cells that communicate with both the neuron and the extracellular milieu). One of the elementary motifs for receptor regulation is shown in the figure.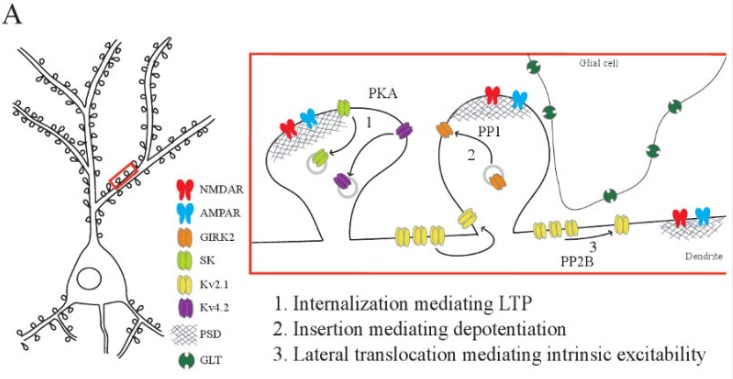 (figure from Shah et al 2010) Neurotransmitter receptors are assembled in the endoplasmic reticulum of neurons, which can be found in dendrites as well as in the cell body. In dendrites, ER is often found near branch points and spines, both of which provide short paths to synapses. After production, the receptor (likely in the form of subunits, which may or may not be active yet) is inserted into the presynaptic membrane around the edges, and migrates into the region of the postsynaptic density, where it attaches itself to the cytoskeleton which anchors it in place. This process is shown in the figures relative to the synaptic densitites.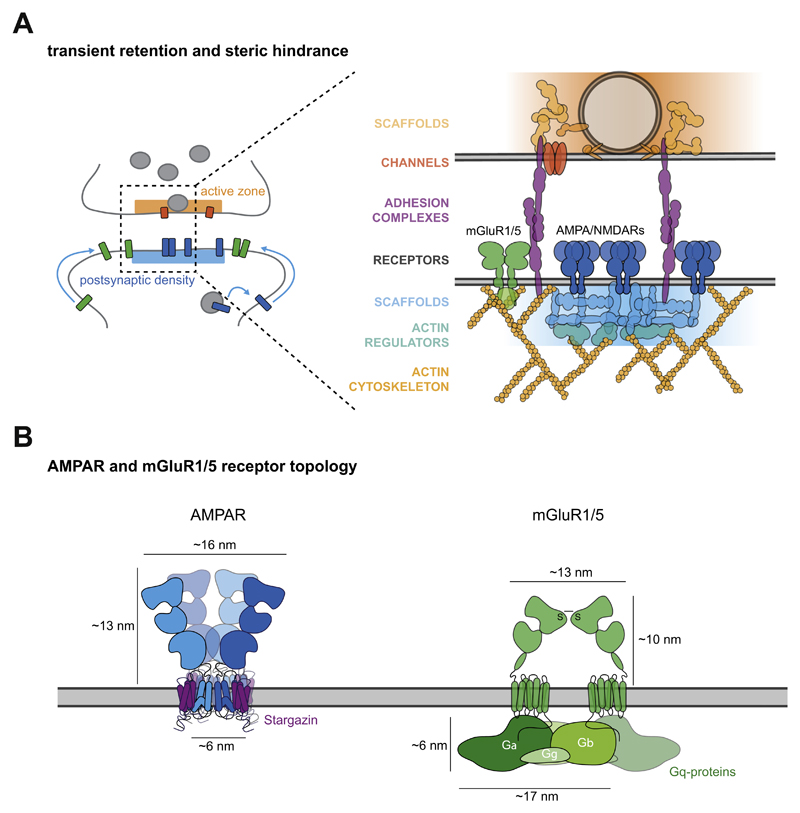 (figure from Scheefhals and MacGillavry 2018) 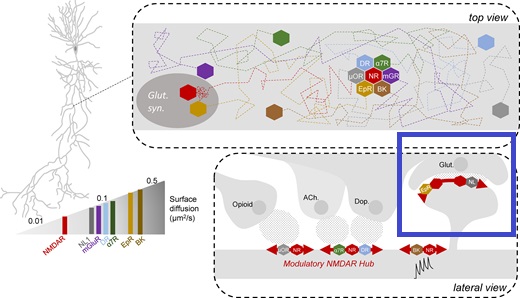 (figure from Petit-Pedrol & Groc 2020) In addition to direct synaptic effects there may be indirect effects at the synapse, mediated by receptors and channels that reside on glial cells. Such interactions are common in tripartite synapses. An example of a tripartite synapse is shown in the figure.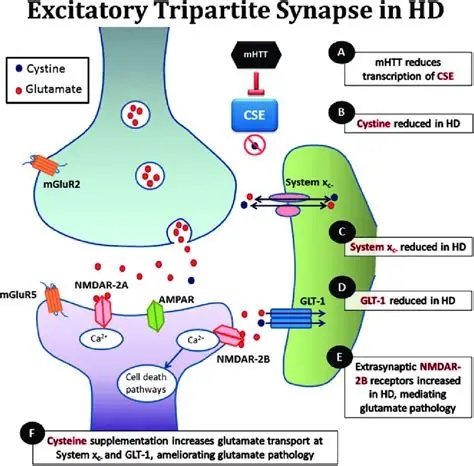 (figure from Wright et al 2016) 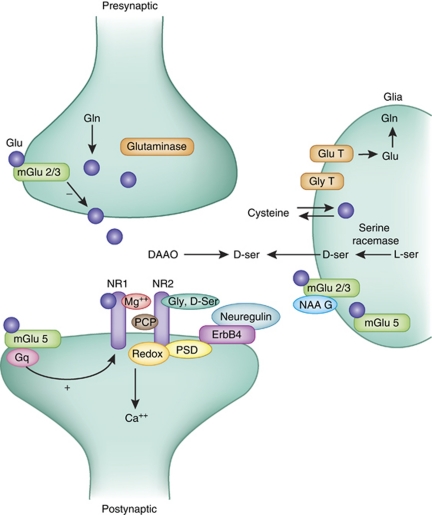 (figure from Moghaddam and Javitt 2011) In the synapse, embedded into the synaptic membranes on each side, are "micro-domains" or "nano-domains", consisting of pockets of receptors and vesicle docking sites. Newly synthesized or recycled receptors that enter the synaptic density along the sides, tend to migrate into the domains, increasing both the frequency of domains and their size. These have differential effects, as shown in the figure. The number of receptors, their placement, and their internal configurations can be controlled through synaptic plasticity.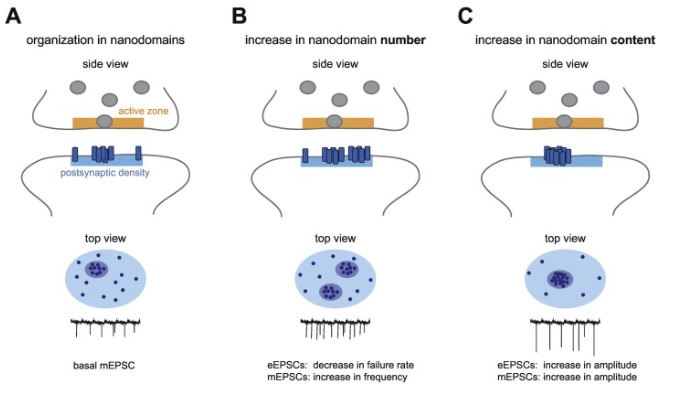 (figure from Scheefhals and MacGillavry 2018) 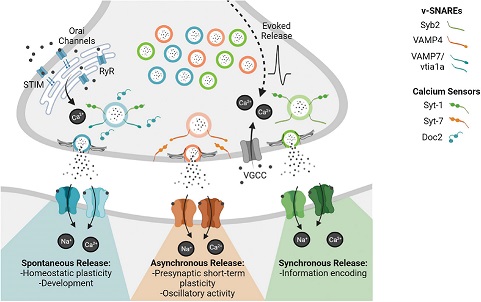 (figure from Guzikowski and Kavalali 2021) Plasticity and ModifiabilityThe transmission strength at a synapse can be modified in many ways. The traditional way of looking at this is in terms of a synaptic "weight", an ancient nomenclature that came out of the Perceptron era. Sometimes, the weights are modified on the basis of incoming data, and at other times, they are controlled by internal processes (like during sleep, when brain activity changes considerably). Plasticity can be achieved presynaptically, postsynaptically, or both - and it can also be achieved in conjunction with glial cells, especially at tripartite synapses. 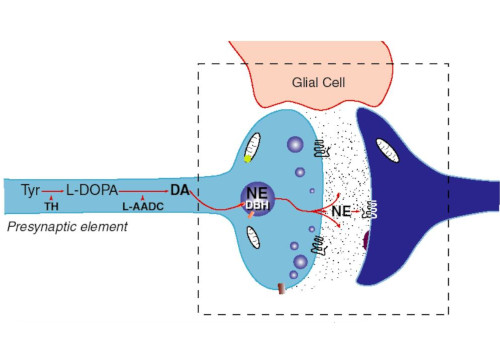 Plasticity is a general term that describes dozens of very specific types of synaptic modifiability. These have different time courses, related to differing chemical kinetics and reaction rates. Historically, all the different kinds of plasticity depend on temporal correlations between the pre- and post-synaptic neurons. This is the Hebbian model, named after psychologist Donald Hebb (although he didn't invent it, it dates back to the middle 19th century). Hebbian plasticity may include ordinary adaptation, facilitation and depression, PTP, STP, STD, LTP, LTD, STDP, and a bewildering array of additional types of plasticity (please refer to the glossary for the definitions of these terms). We'll just introduce the concept of plasticity here, and discuss its various forms and their uses in more detail in a separate section. 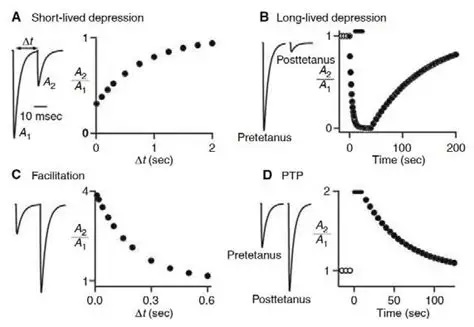 The time course of long term plasticity induced by a pairing protocol is shown in the next figure. Once again, please note the time courses.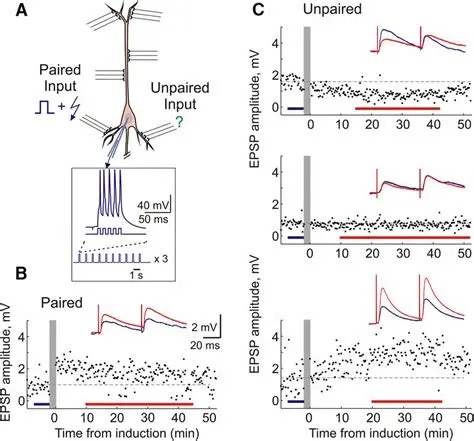 (figure from Bannon et al 2016) In addition to these unidirectional modifications there are also bidirectional modifications. For example in STDP (spike timing-dependent plasticity), the modification can be either potentiation or depression, depending on whether the presynaptic signal occurs before or after the postsynaptic spike. This form of plasticity is shown in the figure.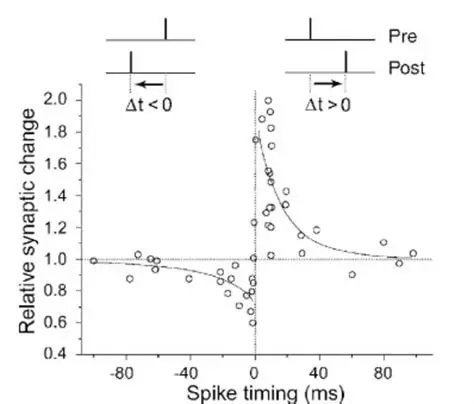 (figure from Bi & Poo 1998) Spines and Spine ApparatusThe mechanisms of neurotransmission around a spiny synapse may be considerably more complicated than those around ordinary aspiny synapses. A molecular "spine apparatus" is often found in the neighborhood of dendritic spines, beginning with endoplasmic reticulum in the dendritic shaft that inserts into the spine stalk and attaches to the cytoskeleton. The spine apparatus changes size and shape, as does the spine itself. It is likely that the spine apparatus is involved in the local control of neurotransmission, including specifically receptor levels and postsynaptic plasticity.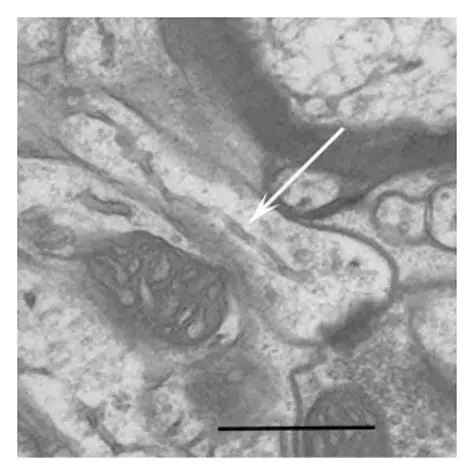 (figure from Jasinska et al 2016) Neuro-ModulationThere are synapses from axons to dendrites, from dendrites to dendrites, and from axons to other axons. Many of these synapses don't alter the membrane potential directly, instead they control the transmission through another synapse, or another portion of the same synapse. These "modulators" are ubiquitous in the brain, and many of them are associated with well known transmitters like acetylcholine, dopamine, and serotonin. There are modulators that act at the level of the neuron, and modulators that act at the level of individual synapses. Axo-axonic synapses are examples of the latter, the outer synapse will typically modulate the activity of the inner synapse. The nature of the modulation can vary greatly. In some cases it is associated with underlying plasticity, but in many cases the effect of neuromodulation is temporary and short lived. The figures show an axodendritic synapse, an axo-axonic synapse, and a dendro-dendritic synapse. The axon terminals have plenty of vesicles, the dendrites don't.   Neuromodulation is affected by many of the same regulatory mechanisms that control synapses. For example one of the side effects of opiate addiction is "supersensitivity", where the amount of endogenous receptor has been down-regulated to accommodate the presence of the drug. Normally, endogenous opiates inhibit transmission through the pain pathways, by a variety of mechanisms including G-coupling to increase intracellular cGMP and a calcium mediated effect on an inwardly rectifying potassium conductance. Upon rebound, there are not enough receptors to support the resumption of normal inhibitory control over the pain pathways, and the results are unpleasant, until the proper setpoint can be achieved again, over the course of several days.Next we'll take a look at some of the network behaviors that can be modified by synapses. Among the most important of these are rhythmic behaviors, which go beyond a simple algebraic manipulation of the weight matrix.Next: Rhythmic Behavior |