Neurons are biophysical devices, they perform computations and transmit signals with combinations of biophysical behaviors. Biological environments are always stochastic and noisy, very little is deterministic about them. The behaviors are constrained dynamically by coupling among related systems. The membrane potential of neurons is a great case study, in this regard.
The careful management of ion concentrations inside and outside the cell leads to electrical behavior that can be used for computations. (There is more to computation than just electrical behavior, but it's a good place to start). Most relevant are the monovalent ions like sodium, potassium, and chloride (and even protons, H+ ions), and positively charged divalent cations (calcium, magnesium) play an important role in many neurons and glia. The behavior of a given neuron is determined in large part by how the ions are managed. There are ion "channels", and there are also ion "pumps". Some of these are voltage dependent, others are passive.
From research into actual neurons, it becomes clear that simplistic neural models are inadequate to describe biological reality. A spiking model like integrate-and-fire is better than a linear model like McCulloch-Pitts, but none of these do justice to the full scope of biological behavior. The inadequacies can be seen in things as simple as plateau potentials, which so far are mostly missing from the synthetic repertoire.
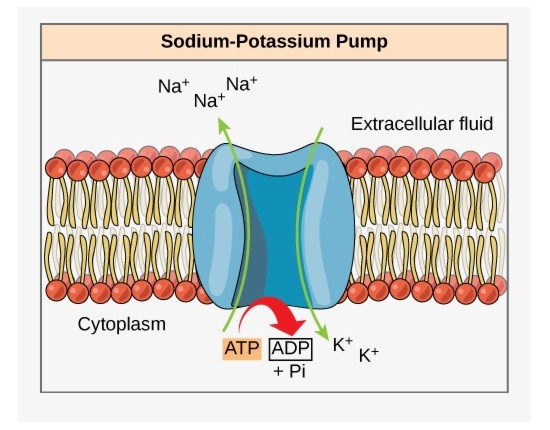
The Sodium PumpThe neural membrane potential begins with an active transmembrane protein that pumps sodium ions out of the cell. The result is that there are more positively charged sodium ions outside the cell than inside, and the result is a transmembrane potential, a difference in charge between the inside and outside. The protein molecule is loosely called a "sodium pump", and in reality there are several varieties of these pumps, they're built from protein subunits that are assembled in different ways. In any case, the sodium ion concentrations inside and outside the cell will eventually reach equilibrium with the pumping process, and the resulting membrane potential can be calculated using the Nernst equation, which calculates the equilibrium potential of an ion distribution. The equilibrium potential is dependent on the ratio of ions inside the cell to outside.

For monovalent cations like sodium and potassium this works out to V (mV) = 58 log [Cout]/[Cin] where V is the equilibrium potential and C is the ion concentration. The equilibrium potential is temperature dependent, but in biological systems the temperature is carefully regulated and consistent.
In most human brain tissue the sodium pump is coupled to a potassium pump, that shuttles potassium ions the other way, from outside the cell to inside. Each cycle of the coupled sodium-potassium pump sends three sodium ions out, and allows two potassium ions in (this occurs at the expense of one energy-providing ATP molecule). The net result is still an efflux of positive charge. This process of shuttling ions back and forth in an energy-driven manner is called active transport.
Potassium and Chloride ChannelsIn addition to active pumping, there are also passive channels that simply allow ions to travel across the cell membrane. Examples of these include passive potassium and chloride conductances. Ions that can freely diffuse across the membrane will arrange themselves according to voltage and concentration gradients. When there are already gradients set up by an active sodium pump, the total effect of other ions on the membrane potential is given by the Goldman equation, also known as the Goldman-Huxley-Katz equation or GHK.

The relative permeabilities of each ion across the membrane (P above) determine their contributions to the final resting potential.
In many cases these ion conductances are linked to synaptic receptors, and can be manipulated by synaptic activity. We'll discuss synaptic activity in more detail, for now let's just consider the neuron without synapses. In many neurons, there is an uneven distribution of channels along the axons and dendrites. For example sodium channels may cluster around the axon hillock (the part of the axon nearest the cell body, that initiates the action potential), and calcium channels in particular are found in the dendritic tree and the synaptic terminals but not in the main body of the axon. These distributions affect the behavior of compartments within the neuron. In dendrites especially, there are processing compartments with unique channel distribution signatures, and the regulation of ion channels (both free standing and attached to synaptic receptors) has been extensively studied and remains an area of active research.
Voltage Dependent ConductancesWith just passive conductances, the nerve membrane will smoothly integrate its inputs. Adjustments to the conductances will simply result in graded changes to the membrane potential, according to the GHK equation (and its extensions if there are more ion types or conductance types). However there are also active conductances that are voltage-dependent, and the introduction of such conductances can make the membrane response highly nonlinear, as well as introducing a wide variety of dynamics.
The action potential (commonly called a "spike", when the neuron "fires") is enabled by a voltage dependent sodium channel, that only opens up when the membrane potential reaches a threshold value. The threshold value is usually more positive than the resting potential. The resting potential is typically in the range of -80 to -50 mV, while the threshold is typically in the range of -50 to -20 mV. Sometimes there is a small contribution to the threshold from charged atoms within the channel, this contribution is called a "gating current" and it can be experimentally separated from the ion current. The figure shows a voltage gated sodium conductance.
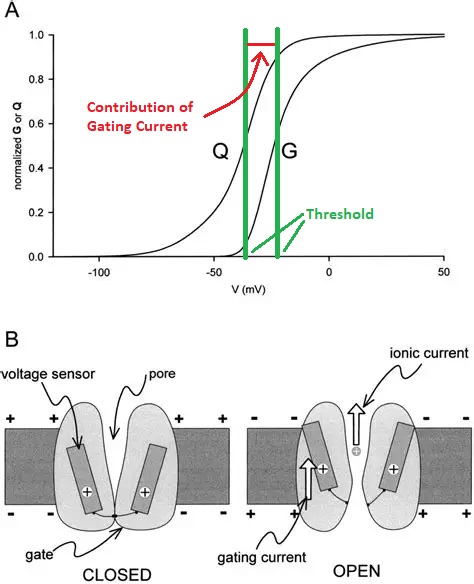
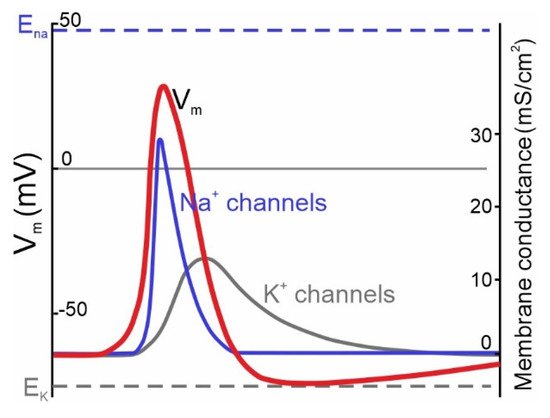
The action potential is regenerative, it has a positive feedback dynamic. The activation of the voltage dependent sodium current (by a supra-threshold membrane potential) causes sodium ions to rush into the cell (along their voltage and concentration gradient), making the membrane potential more positive, opening up even more voltage-gated sodium channels. This causes the membrane potential to temporarily go positive, and at some point it will typically overshoot 0, before other currents turn it back off. Usually it is the passive potassium current that restores the membrane potential, as potassium ions cross the membrane in relation to the new voltage gradient.
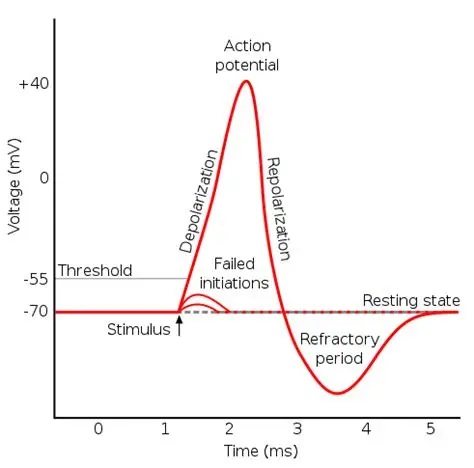
Following an action potential there is typically a small undershoot, as shown in the figure, during which the neuron can not fire again (because its ion supplies need to be replenished for the membrane potential to be restored). The area of undershoot is called the refractory period. Typically the refractory period is on the order of a millisecond or two, so the maximum firing rate of a neuron might be around 1000 Hz. This figure shows the time course of the currents related to the action potential. You can see the delayed potassium current relative to the sodium current.
To turn off the voltage dependent sodium current and end the spike, potassium and chloride ions rearrange themselves to bring the membrane potential back down to its equilibrium value. This turns off the voltage dependent sodium conductance when the membrane potential goes back down below the threshold value. The neuron eventually returns to its resting state, but before acheiving the final resting potential there is usually a small negative overshoot while the potassium and chloride ions are rearranging themselves, which may extend beyond the actual experimentally measured refractory period.
Chloride is particularly interesting because its equilibrium potential is close to the resting potential. When we discuss synaptic transmission, this will lead to synapses that can be either excitatory or inhibitory, depending on the current membrane potential. This capability is very useful for computation, it enables a special kind of neural behavior that goes beyond the traditional pattern of plastic excitatory and inhibitory synapses.
Hodgkin-Huxley EquationsThe Hodgkin-Huxley equations model the membrane potential of a neuron, based on its conductances. They begin with an electrical model because it conveniently represents the currents.
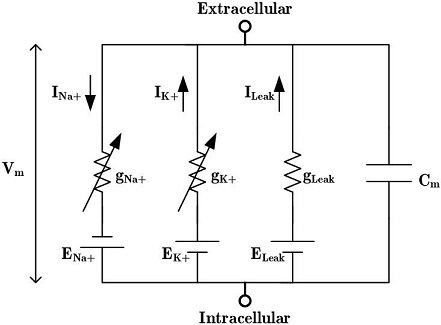
The H-H equations are a set of coupled differential equations relating the currents to the membrane potential.

The parameters m, n, and p in the above equations relate to the subunit composition of the ion channels. Hodgkin and Huxley began by curve fitting their results to polynomials, and discovered these relationships without any specific knowledge of the underlying biochemistry.
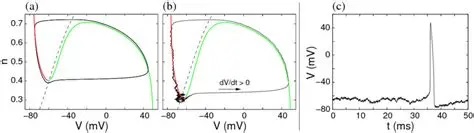
It is helpful to visualize the membrane behavior in phase space, and see how it relates to the voltages that we actually measure with an electrode.
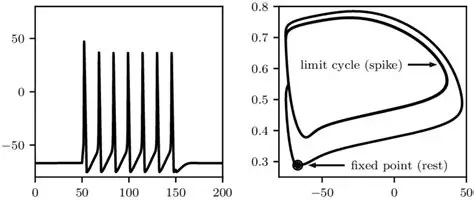
Calcium ChannelsCalcium is a very special molecule in the brain. It's important not only for neurons but also for glia, and it's carefully controlled both inside and outside the cell. Other divalent cations are relevant too, for example magnesium plays an important role in glutamate signaling, but calcium is ubiquitous and it links to a bewildering array of neuronal behaviors.
In neurons, most calcium channels are attached to synapses. When calcium enters the cell it depolarizes the membrane because of its positive charge, but it has many other effects too, including effects on steroids and prostaglandins, the cytoskeleton, and even direct effects on the cell membrane itself. The best known function of calcium in neurons is its involvement in neurotransmitter release. In synaptic terminals, the arriving action potential opens up voltage-dependent calcium channels that cause transmitter-filled vesicles to fuse with the presynaptic membrane and release their contents into the synaptic cleft. This action of calcium is especially important in the many kinds of synapses that use glutamate as an excitatory neurotransmitter. In such synapses, multiple receptor types are often found side by side, and even on adjoining glial cells.
In dendrites, calcium channels are frequently found in the synaptic compartments called "spines", which are tiny mushroom shaped extrusions with thin stalks that host synapses. Also in dendrites, there may be voltage dependent calcium channels, and dendritic segments can sometimes generate small action potentials called "dendritic spikes". These are common in spiny neurons, they can be found in the cerebral cortex, cerebellum, hippocampus, basal ganglia, and many other places in the brain. A dendritic spike does not necessarily lead to an action potential in the axon, it may dissipate or be inhibited before it gets there. (Dendritic spikes are also associated with bistability in the membrane potential at the neuronal cell body, which we'll talk about in the next section on synapses).
Calcium is heavily regulated by glial cells, especially astrocytes. There are three times as many glial cells in the brain as there are neurons. They are linked by gap junctions (they're electrically conductive), and they interact with neurons chemically, which means they can affect electrical computation indirectly. One of the ways astrocytes regulate their surroundings is by regulating calcium concentrations, both in the extracellular milieu and in synapses. Astrocytes frequently have processes that wrap themselves around synapses, and they're often found around glutamatergic synapses that are calcium-sensitive. Astrocytic calcium waves can be visualized after neural electrical stimulation (YouTube has some great videos), and the resulting behavior modeled at both the electrical and chemical level (Musotto et al 2025).
Calcium is especially important in neurons because it's related to sub-threshold membrane potential oscillations even in the absence of input. These subthreshold oscillations are properties of the individual neurons, they do not depend on population interactions (Zhuchkova et al 2013, Amarillo et al 2014). There are a number of potential sources of periodic behavior in a cell, not the least of which are the microtubules which have a resonance in the 39 Hz range (Cantero and Cantiello 2020, Mohsin et al 2025). The figure shows a very simple model containing only leak and calcium currents, resulting in subthreshold oscillations in the 10 Hz range.
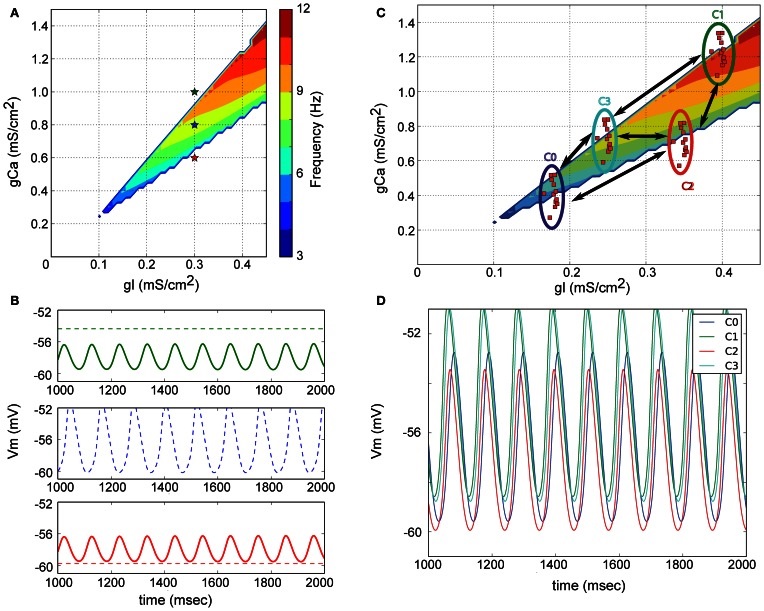
(figure from Lefler et al 2013 Creative Commons)
Such subthreshold activity is important because it interacts with other periodic behavior in the network, as we'll see on a subsequent page when we discuss modulation. Neurons inhibited by the neurotransmitter GABA often display rebound bursting after the inhibition, and the frequency of action potentials inside the burst is related to the frequency of subthreshold oscillations. And, astrocytes add additional dynamic modes to neuronal systems (Palabas et al 2023).
The dynamic of the Hodgkin-Huxley equation is four dimensional and difficult to visualize that way. The figure shows the phase plane reduced to two dimensions. We'll talk about ways of simplifying the equations in the section on model neurons. An important concept in modeling is preserving the "essential behavior", and what is essential depends somewhat on the model, however in the case of neurons the spiking behavior can certainly be considered essential, and there are a number of computationally accessible ways to accomplish it.
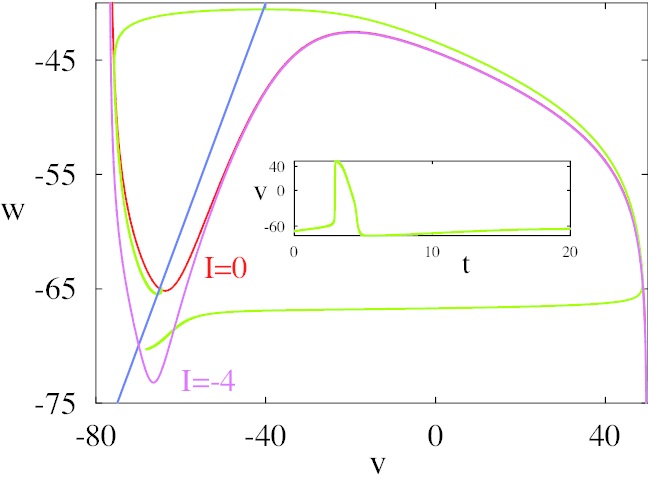
(figure from Chik et al 2004)
Next: Synapses |











